Bisdisulizole disodium
 | |
 | |
| Names | |
|---|---|
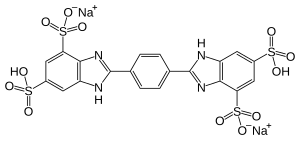
| Preferred IUPAC name Disodium 2,2′-(1,4-phenylene)bis(6-sulfo-1H-1,3-benzimidazole-4-sulfonate) | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.121.951 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C20H12N4Na2O12S4 |
| Molar mass | 674.55 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Bisdisulizole disodium (INN[1]/USAN,[2] trade name Neo Heliopan AP, INCI disodium phenyl dibenzimidazole tetrasulfonate) is a water-soluble organic compound which is added to sunscreen products to absorb UVA rays. It is marketed by Symrise.
The use of bisdisulizole disodium has been approved in the European Union (since the year 2000)[3] and other parts of the world up to 10% in sunscreen formulations,[4][5][6] while it is not approved in the United States.
References
- ^ http://ec.europa.eu/enterprise/cosmetics/cosing/index.cfm?fuseaction=search.detailsPDF&id=75792[permanent dead link]
- ^ "Statement on a nonproprietary name adopted by the USAN Council: Bisdisulizole disodium" (PDF). American Medical Association. April 4, 2006. Retrieved 2008-01-08.
- ^ Osstervalder U, Luther H, Herzog B (2001). "Über den Lichtschutzfaktor hinaus - neue effiziente und photostabile UVA-Filter". Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (in German). 44 (5): 463–70. doi:10.1007/s001030170019. S2CID 36985446.
- ^ "10. Sunscreens" (PDF). Australian regulatory guidelines for OTC medicines. Commonwealth Department of Health and Ageing, Therapeutic Goods Administration. July 1, 2003. p. 14. Archived from the original (PDF) on August 31, 2007. Retrieved 2008-01-08.
- ^ "Symrise UV protection products" (PDF). Symrise. Archived from the original (PDF) on 2008-08-21. Retrieved 2008-08-10.
- ^ "Cosmetic Products Regulation, Annex VI - Allowed UV Filters". ECHA. Retrieved 2023-08-15.
- v
- t
- e
Sunscreening agents approved by the US FDA or other agencies
- UVA: 400–315 nm
- UVB: 315–290 nm
- Chemical agents unless otherwise noted
- Avobenzone (Parsol 1789)
- Bisdisulizole disodium (Neo Heliopan AP)
- Diethylamino hydroxybenzoyl hexyl benzoate (Uvinul A Plus)
- Ecamsule (Mexoryl SX)
- Menthyl anthranilate
- Amiloxate
- 4-Aminobenzoic acid (PABA)
- Cinoxate
- Ethylhexyl triazone (Uvinul T 150)
- Homosalate
- 4-Methylbenzylidene camphor (Enzacamene)
- Octyl methoxycinnamate (Octinoxate)
- Octyl salicylate (Octisalate)
- Padimate O (Escalol 507)
- Phenylbenzimidazole sulfonic acid (Ensulizole)
- Polysilicone-15 (Parsol SLX)
- Trolamine salicylate
 | This dermatologic drug article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e










